Contributed by Hala Karnib, M.D.1, Jamie Sheth, M.D. 2, Anita Valanju Shelgikar, M.D.1 1Sleep Disorders Center, Department of Neurology, University of Michigan, Ann Arbor, MI; 2Division of Pulmonary & Critical Medicine, Department of Internal Medicine, University of Michigan, Ann Arbor, MI Corresponding Author: Hala Karnib, M.D., Michael Aldrich Sleep Disorders Laboratory, Med Inn, Box 5845, 1500 East Medical Center Drive, Ann Arbor, MI 48109-5845; Tel: (734) 232-4600; Fax: (734) 647-9065; Email: halaK24@gmail.com Disclosures: This was not an industry supported study. The authors have no commercial conflicts of interest to disclose. MeSH Key Words: obstructive sleep apnea; central sleep apnea with Cheyne-Stokes breathing pattern; positive airway pressure; adaptive servoventilation
In Brief
A middle-aged man hospitalized for new onset of severe systolic heart failure requiring mechanical ventilation. Shortly after he was extubated, a caregiver witnessed episodes of apnea with associated oxygen desaturation at night. Polysomnography revealed severe sleep disordered breathing that was abolished with treatment.
Case Vignette
A 46-year-old man presented with increased dyspnea and fatigue over several months, which worsened acutely before his presentation to an Emergency Department. He was an avid body builder and gave a history of alcohol and anabolic steroid abuse.
The man was admitted to the intensive care unit for management of severe systolic heart failure with ejection fraction 10-15% and cardiogenic shock with hypoxic respiratory failure requiring intubation. Further evaluation lead to a diagnosis of chronic non-ischemic cardiomyopathy due to alcohol abuse and anabolic steroid use. The acute decompensation was attributed to viral myocarditis, given the history of recent upper respiratory infection.
Heart failure was treated with diuresis and afterload reduction. He was euvolemic at the time of extubation. Shortly afterwards, a caregiver observed episodes of apnea during sleep with oxygen desaturations into the 80’s. Additional history from the patient’s family revealed a long-standing history of snoring and significant use of abdominal muscles during sleep, but no witnessed apneas prior to the current hospitalization. The patient reported that, prior to admission, he often took daily "catnaps" lasting approximately 30 minutes. He denied drowsiness while driving. An Epworth Sleepiness Scale was not calculated.
The patient was seen and evaluated by a sleep medicine physician 11 days after initial presentation (4 days after extubation). When the patient was awake, arterial oxygen saturation was in the low 90’s while he breathed ambient air. The patient appeared euvolemic and intermittently dozed with significant snoring. Body mass index was 30 kg/m2. Neck circumference was 17.5 inches. Oral airway was crowded (modified Mallampatti class IV) with slight retrognathia and a large tongue.
Ejection fraction improved from less than 10% to 20% on repeat echocardiogram. An inpatient split-night polysomnogram was performed (Table 1; Figures 1 and 2) followed the next night by a retitration study (Figure 3).
| Respiratory Analysis: | Baseline |
|---|---|
| Apneic Episodes (#) | 21 |
| Total Sleep time (min) | 15.5 |
| Supine (min) | 15.5 |
| Apnea/Hypopnea Index† (AHI) | 86.7 |
| NREM AHI | 86.7 |
| REM AHI | - |
| Respiratory Disturbance Index | 86.7 |
| Obstructive Apnea Index | 31.0 |
| Central Apnea Index | 53.7 |
| Hypopnea Index | 0.0 |
| RERA Index | 0.0 |
| Mean Sleep % SpO2 | 88 |
| Min % SpO2 | 80 |
†Index = no./hour of sleep
Table 1. Baseline Polysomnogram, respiratory analysis.
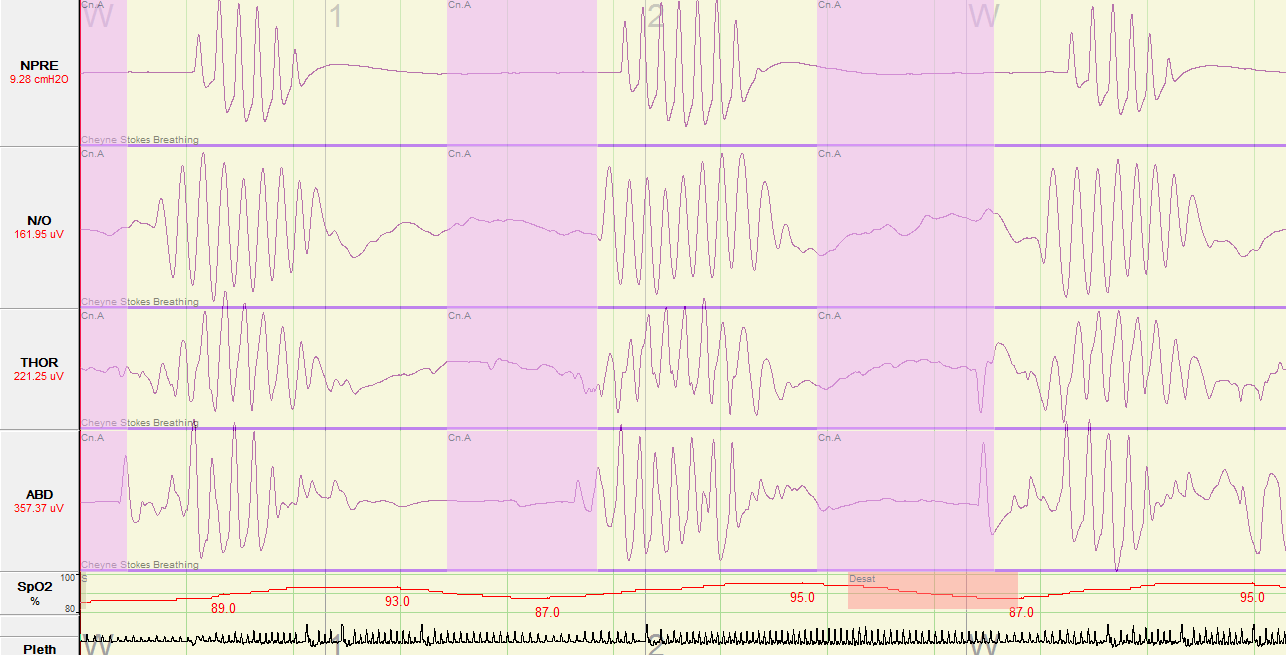
Figure 1. Baseline Polysomnogram. A Type I polysomnogram was performed. In order to better illustrate the respiratory events the following channels were selected for display: NPRE=nasal pressure; N/O=nasal/oral thermistor; THOR=thoracic respiratory effort; ABD=abdominal respiratory effort; SpO2=Peripheral capillary oxygen saturation; Pleth=plethysmography.
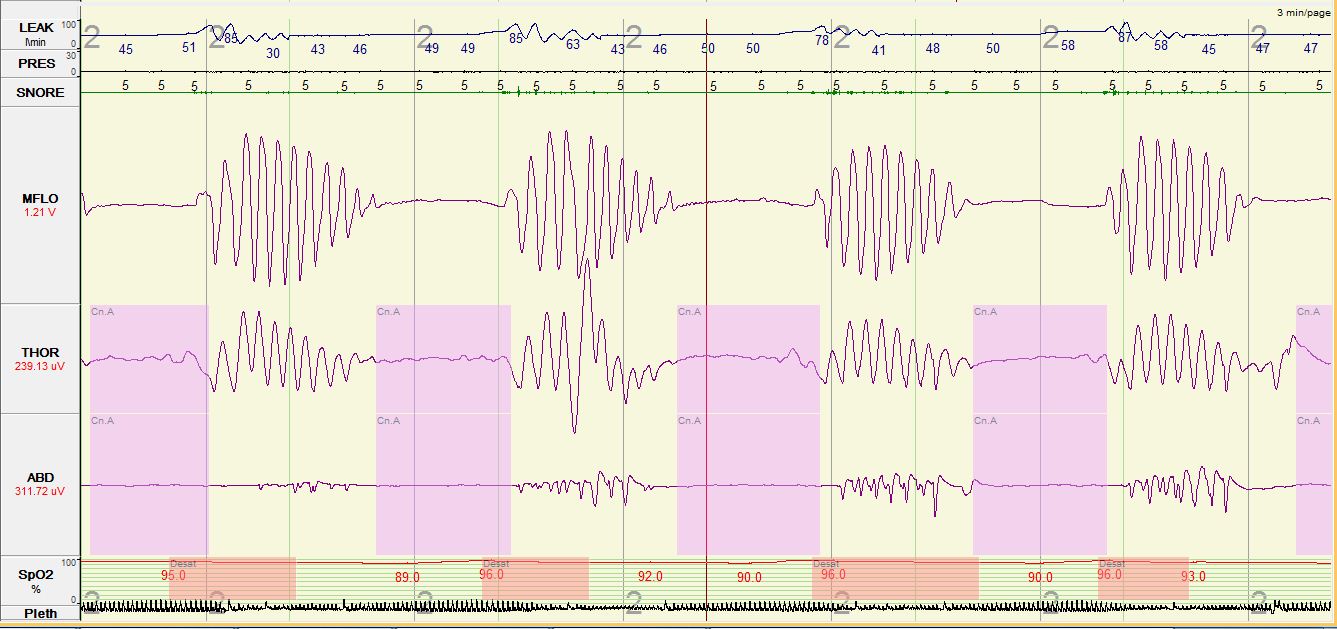
Figure 2. Please note, a Type I polysomnogram was performed. In order to better illustrate the respiratory events, the following channels were selected for display: Leak=mask leak; PRES=positive airway pressure setting; MFLO=mask flow; THOR=thoracic respiratory effort; ABD=abdominal respiratory effort; SpO2=Peripheral capillary oxygen saturation; Pleth=plethysmography.
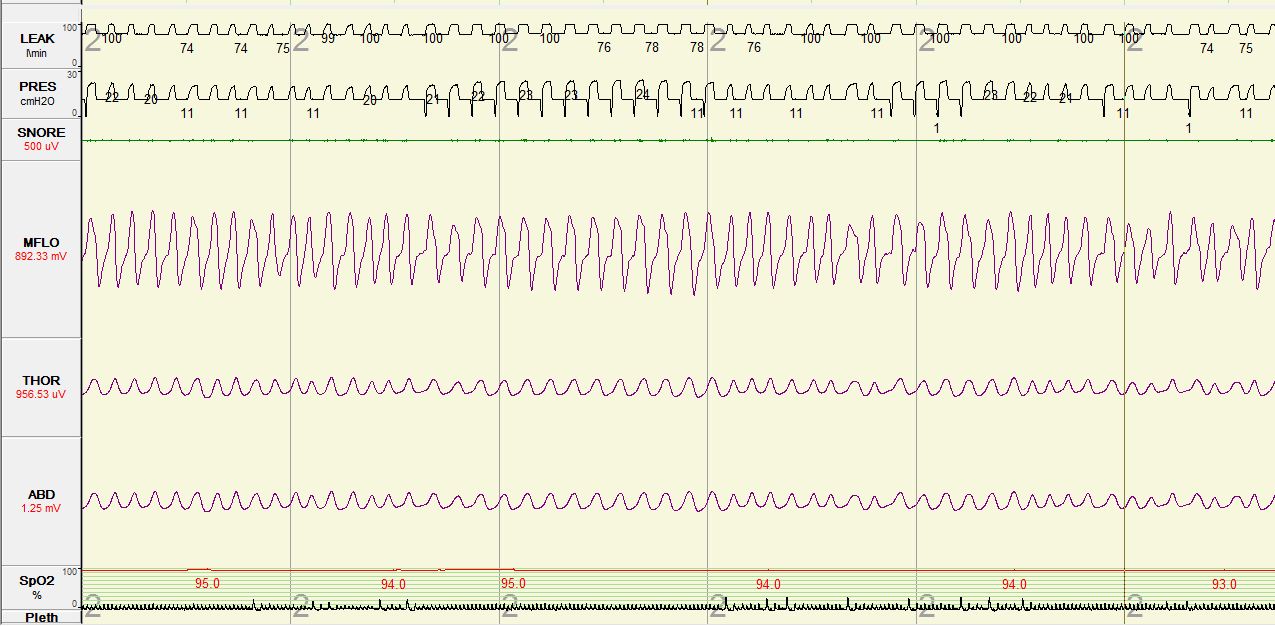
Figure 3. Adaptive Servoventilation (ASV) Titration. A Type I polysomnogram was performed. In order to better illustrate the respiratory events, the following channels were selected for display: Leak=mask leak; PRES=positive airway pressure setting; MFLO=mask flow; THOR=thoracic respiratory effort; ABD=abdominal respiratory effort; SpO2=Peripheral capillary oxygen saturation; Pleth=plethysmography.
Questions
-
What is the apparent diagnosis?
-
What are risk factors for this type of sleep-disordered breathing pattern?
-
What treatment options may be considered for this sleep-disordered breathing pattern?
Discussion
The split-night polysomnogram showed a cyclic crescendo-decrescendo breathing pattern consistent with severe central sleep apnea in a Cheyne-Stokes breathing pattern and arterial oxygen desaturations to the low to mid 80’s (Figure 1). The titration portion tested continuous positive airway pressure (CPAP) and bi-level positive airway pressure (BPAP); central hypopneas persisted though sleep consolidation improved. Re-titration with adaptive servoventilation was associated with increased sleep consolidation, baseline oxygen saturation of 94-97%, and resolution of central apneas, central hypopneas and Cheyne-Stokes respirations (Figure 3).
Central sleep apnea (CSA) is mediated by central and peripheral chemoreceptors leading to respiratory events that are characterized by decreased flow as a result of decreased or absent respiratory effort. Central sleep apnea can be further sub-categorized into primary central sleep apnea, central sleep apnea with Cheyne-Stokes breathing (CSA-CSB) pattern, central sleep apnea due to high-altitude periodic breathing, central sleep apnea due to medical disorder without Cheyne-Stokes Breathing, and central sleep apnea due to a medication or substance (1). A thorough clinical history and understanding of the risk factors associated with different forms of sleep-disordered breathing can help ensure proper diagnosis and management.
Central sleep apnea, specifically CSA-CSB, is associated with certain medical conditions including heart failure, renal failure and stroke (1). Cheyne-Stokes breathing is defined as three or more central respiratory events separated by a crescendo and decrescendo change in breathing amplitude with a cycle length of over 40 seconds; in addition, five or more central respiratory events per hour of sleep associated with crescendo/decrescendo breathing pattern must be recorded over two hours of monitoring (Figure 1) (2). The prevalence of CSA-CSB among patients with systolic heart failure is estimated to be 25-40% (3). In addition to the typical subjective complaints and physiologic consequences that sleep-disordered breathing may cause, CSA-CSB has been associated with increased mortality particularly in the heart failure population (4, 5). Risk factors for central sleep apnea in heart failure include male sex; higher New York Heart Association functional class; lower ejection fraction; waking hypocapnia, defined as arterial partial pressure of carbon dioxide (PaCO2) <38 mm of Hg; higher prevalence of atrial fibrillation; higher B-type natriuretic peptide levels; and frequent nocturnal ventricular arrhythmias (6).
The mechanisms that drive central sleep apnea with Cheyne-Stokes breathing pattern in heart failure patients are interesting and complex. The respiratory cycle is influenced by many factors including peripheral and central chemoreceptors, intrapulmonary receptors and chest wall and muscle mechanoreceptors (7). The primary mediator of ventilation is PaCO2 via central chemoreceptors. Heart failure patients have a tendency to hyperventilate, which drives PaCO2 values below the apneic threshold. Subsequently, apneic episodes lead to an elevated PaCo2 value, which in turn drives hyperventilation. This results in a cyclic, crescendo-decrescendo pattern.
First line treatment of central sleep apnea in patients with acutely decompensated heart failure should be optimization of heart failure. However, if symptoms persist despite optimal medical therapy, diagnostic studies and other treatment options should be considered. In our patient, an inpatient polysomnogram was considered due to excessive daytime sleepiness that impeded his participation in rehabilitation therapy and to high clinical suspicion of obstructive sleep apnea. The baseline portion of his polysomnogram showed sleep-disordered breathing predominately characterized by central sleep apnea with Cheyne-Stokes breathing pattern. Recommendations on when to treat sleep-disordered breathing in the setting of acutely decompensated heart failure are not available. However, a few studies have found that in patients hospitalized with acute systolic heart failure the presence of sleep-disordered breathing was an independent risk factor for readmission and increased mortality (8, 9).
Currently, the American Academy of Sleep Medicine practice parameters recommend continuous positive airway pressure (CPAP), adaptive servoventilation (ASV) and nocturnal oxygen as standard treatments for of central sleep apnea in patients with heart failure. Bi-level PAP with spontaneous/ timed mode may be considered for treatment only if there is no response to the aforementioned treatment options (10).
In post hoc analysis of the CANPAP trial, Arzt et al. reported that when central sleep apnea is effectively treated (defined as AHI < 15) CPAP has a positive effect on transplant free survival and left ventricular ejection fraction (LVEF) (11). Although the data are limited, multiple small studies have shown that adaptive servoventilation (ASV) is better tolerated by heart failure patients. By delivering preset minute ventilation and a backup respiratory rate, ASV normalizes the breathing pattern via reduction of hyperventilation and associated hypocapnia. ASV has also been shown to improve sleep-disordered breathing, LVEF, and compliance, though large-scale randomized trials are needed to further examine these outcomes (12, 13, 14).
The SERVE-HF study is a multinational, multicenter, randomized controlled Phase IV study on the effect of treatment with ASV on morbidity, mortality, and cost-effectiveness ASV therapy, in patients with chronic heart failure and predominantly CSA-CSB (15). In this trial, patients with chronic, symptomatic heart failure (NYHA class 2-4) with ejection fraction ≤ 45 % and moderate to severe central sleep apnea (defined as an AHI >15, >50% of the events being central in nature, and central apnea index at least 10) were randomized to two arms: best medical care alone versus best medical care plus ASV titrated to minimize AHI.
On May 13, 2015, ResMed released a safety notice regarding the use of ASV in heart failure patients based on the preliminary results of the SERVE-HF study (16). Preliminary analysis of the data showed a statistically significant increase in absolute risk of cardiovascular mortality for patients in the arm that received ASV compared to those in the control group (2.8% absolute increased risk). Of note, this study cannot be applied to patients with heart failure and other forms of central sleep apnea. Results of a final analysis, including sub-group analyses, are still pending.
The American Academy of Sleep Medicine has recommended (17): 1. ASV not be prescribed to treat central sleep apnea in patients with symptomatic heart failure and LVEF<45% until further data are available; 2. Clinical assessment for heart failure, along with objective assessment of left ventricular ejection fraction if heart failure symptoms are present, should be done if a patient is started on ASV therapy; 3. Prior patients with symptomatic heart failure treated with ASV since 2005 should be advised of the risks of therapy with the recommendation to discontinue ASV therapy and use another treatment modality.
The mechanism as to why ASV was associated with increased mortality in this group of patients is still unclear. We question the affects of changes in intra-thoracic pressure on heart function. Some have also questioned whether Cheyne-Stokes breathing may in fact be protective in patients with severe congestive heart failure. Although these theories are interesting, the true mechanism remains a mystery and further investigation is needed.
In conclusion, at this pivotal point in our understanding of the management of central sleep apnea and Cheyne-Stokes breathing in patients with systolic heart failure, careful clinical evaluation needs to be considered prior to the initiation of a treatment modality.
Answers
-
Central sleep apnea with Cheyne-Stokes breathing.
-
Cheyne Stokes breathing is commonly associated with cardiac disease (i.e. heart failure). Other associations include neurological disease, renal failure, sedation, normal sleep, acid-base disturbances, prematurity, and altitude acclimatization.
-
Cheyne Stokes respiration can be treated with continuous positive airway pressure (CPAP), adaptive servoventilation (ASV), and nocturnal oxygen.
Follow-Up
Our patient was started on adaptive servoventilation and advised to follow up in Sleep Clinic after discharge from the hospital.
On return 1 month later, cardiovascular evaluation revealed a improvement in left ventricular ejection fraction to 25-30%. He did not return subsequently to our medical center after transfer of care other practices because of limitations in health insurance coverage.
If the patient’s left ventricular ejection fraction does not recover to greater than 45%, an alternative adaptive servoventilation should be explored for the reasons cited in the discussion. Alternative treatment options include supplemental oxygen, CPAP or CPAP with oxygen, although the long term effects of these modalities is unknown. Additionally, optimal treatment of underlying heart failure should continue to be a priority (18).
References
-
American Academy of Sleep Medicine. International classification of sleep disorders, 3rd ed. Darien, IL: American Academy of Sleep Medicine, 2014.
-
Berry B, Brooks R, Gamaldo C, Harding S, Lloyd R, Marcus C, Vaughn B. The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications. Version 2.1
-
MacDonald M, Fang J, Pittman SD, et al. The current prevalence of sleep disordered breathing in congestive heart failure patients treated with beta-blockers. J Clin Sleep Med 2008;4:38–42.
-
Hanly PJ, Zuberi-Khokhar NS. Increased mortality associated with Cheyne-Stokes respiration in patients with congestive heart failure. Am J Respir Crit Care Med. Jan 1996;153(1):272-6.
-
Luo Q, Zhang HL, Tao XC, Zhao ZH, Yang YJ, Liu ZH. Impact of untreated sleep apnea on prognosis of patients with congestive heart failure. Int J Cardiol. Apr 2 2009
-
Khayat R, Small R, Rathman L, et al. Sleep disordered breathing in heart failure: identifying and treating an important but often unrecognized comorbidity in heart failure patients. J Cardiac Fail 2013;19:431–44.
-
Caruana-Montaldo B, Fleeson K, Zwillich CW. The control of breathing in clinical practice. Chest 2000; 117:205-25
-
Khayat R, Abraham W, Patt B, Brinkman V, Wannemacher J, Porter K, Jarjoura D. Central sleep apnea is a predictor of cardiac readmission in hospitalized patients with systolic heart failure. J Card Fail 2012;18: 534e540.
-
Ohmura T, Iwama Y, Kasai T, Kato T, Suda S, Takagi A, Hiroyuki D. Impact of Predischarge Nocturnal Pulse Oximetry (Sleep-Disordered Breathing) on Postdischarge Clinical Outcomes in Hospitalized Patients With Left Ventricular Systolic Dysfunction After Acute Decompensated Heart Failure. The American Journal of Cardiology 2014; 113:697-700. doi:10.1016/j.amjcard.2013.10.048
-
Aurora RN, Chowdhuri S, Ramar K, Bista SR, Casey KR, Lamm Cl, Kristo DA, Mallea JM, Rowley JA, Zak RS, Tracy SL. The treatment of central sleep apnea syndromes in adults: practice parameters with an evidence-based literature review and meta-analyses. SLEEP 2012;35(1):17-40.
-
Arzt M, Floras J, Logan A, et al. Suppression of central sleep apnea by continuous positive airway pressure and transplant-free survival in heart failure: a post hoc analysis of the Canadian continuous positive airway pressure of patients with central sleep apnea and heart failure trial (CANPAP). Circulation 2007;115:3173-80.
-
Philippe C, Stoïca‐Herman M, Drouot X, et al. Compliance with and effectiveness of adaptive servoventilation versus continuous positive airway pressure in the treatment of Cheyne‐Stokes respiration in heart failure over a six month period. Heart 2006; 92(3):337-. doi:10.1136/hrt.2005.060038.
-
Kasai T, Usui Y, Yoshioka T, et al. Effect of flow-triggered adaptive servoventilation compared with continuous positive airway pressure in patients with chronic heart failure with coexisting obstructive sleep apnea and Cheyne-Stokes respiration. Circ Heart Fail 2010;3:140-8
-
Sharma BK, Bakker JP, McSharry DG, Desai AS, Javaheri S, Malhotra A. Adaptive servoventilation for treatment of sleep-disordered breathing in heart failure Chest. 2012;142(5):1211-1221. doi:10.1378/chest.12-0815
-
Cowie MR, Woehrle H, Wegscheider K, Angermann C, d'Ortho MP, Erdmann E, Levy P, Simonds A, Somers VK, Zannad F, Teschler H. Rationale and design of the SERVE-HF study: treatment of sleep-disordered breathing with predominant central sleep apnoea with adaptive servo-ventilation in patients with chronic heart failure. Eur J Heart Fail. 2013 Aug;15(8):937-43. doi: 10.1093/eurjhf/hft051.
-
ResMed Press Release. Available at: http://www.resmed.com/us/en/consumer/newsandinformation/news-releases/2015/resmed-provides-update-on-phase-iv-serve-hf-study-of-adaptive-servo-ventilation-therapy.html. Accessed June 17, 2015.
-
American Academy of Sleep Medicine Special Safety Notice. Available at: http://www.aasmnet.org/articles.aspx?id=5562. Accessed June 17, 2015.
-
Ayas NT, Patil SP, Stanchine M, Malhotra A. Treatment of Central Sleep Apnea with Adaptive Servoventilation in Chronic Heart Failure. AM J Respir Crit Care Med. First published online 17 Jun 2015.



