Reviewed By Allergy, Immunology & Inflammation Assembly
Submitted by
Amitesh Agarwal MD
Senior Fellow, Division of Pulmonary and Critical Care Medicine
University of Texas Medical Branch
Galveston, TX
John Kelley MD
Senior Fellow, Division of Allergy and Clinical Immunology
University of Texas Medical Branch
Galveston, TX
William J Calhoun MD FACP, FCCP, FAAAAI, FACAAI
Professor and Vice Chair for Research
Divisions of Pulmonary and Critical Care Medicine, and Allergy and Clinical Immunology, Department of Internal Medicine
University of Texas Medical Branch
Galveston, TX
Submit your comments to the author(s).
History
A 56-year-old Caucasian female is being referred by a primary care physician for evaluation and management of ‘bad asthma’. She has a history of asthma and allergic rhinitis since childhood and she has taken inhaled medications for many years. She was hospitalized 3 times for asthma, most recently 18 months ago, but was never intubated. Her PCP has prescribed prednisone bursts for her 4 times in the last 3 years.
Her asthma symptoms are principally cough and wheezing, with minimal exertional dyspnea. She reports daily asthma symptoms requiring albuterol for relief, and has nocturnal symptoms about three times monthly.
She is a lifetime non-smoker. She does not have a history of GERD and denies reflux symptoms. A twin sister also has asthma, and her daughter has allergic rhinitis.
Current medications are formoterol/budesonide 160/4.5 mcg combination inhaler two puffs twice daily, daily inhaled tiotropium, daily oral montelukast, intranasal fluticasone two sprays in each nostril twice daily and cetirizine occasionally for rhinitis symptoms, most commonly in the spring months.
She reports that she has occasional dysphagia and dysphonia that have responded to local antifungal treatment, but that with meticulous with inhaler and spacer technique, she has had no such episodes for the past 18 months.
Physical Exam
The nasal turbinates are pale with clear mucoid drainage present. There is no conjunctival injection. The oropharyngeal mucosa appears normal.
Examination of the chest reveals good air entry, with minimal adventitious sounds. Forced expiration results in diffuse audible wheezes.
There are no other physical findings of significance.
Lab
CBC demonstrates WBC 12 k/µL, Hgb 12.6 g/dL, and Hct 38%. The differential includes 0.320 K/µL eosinophils.
Basic metabolic panel, HbA1c and random serum glucose are normal.
IgE is elevated at 360 IU/ml. Serum reactivity to a panel of regionally relevant allergens shows positivity for tree pollens, grass pollens, and both common house dust mites.
FeNO is 18 ppm.
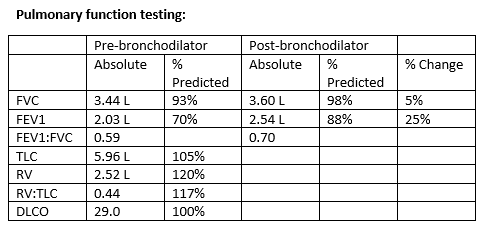
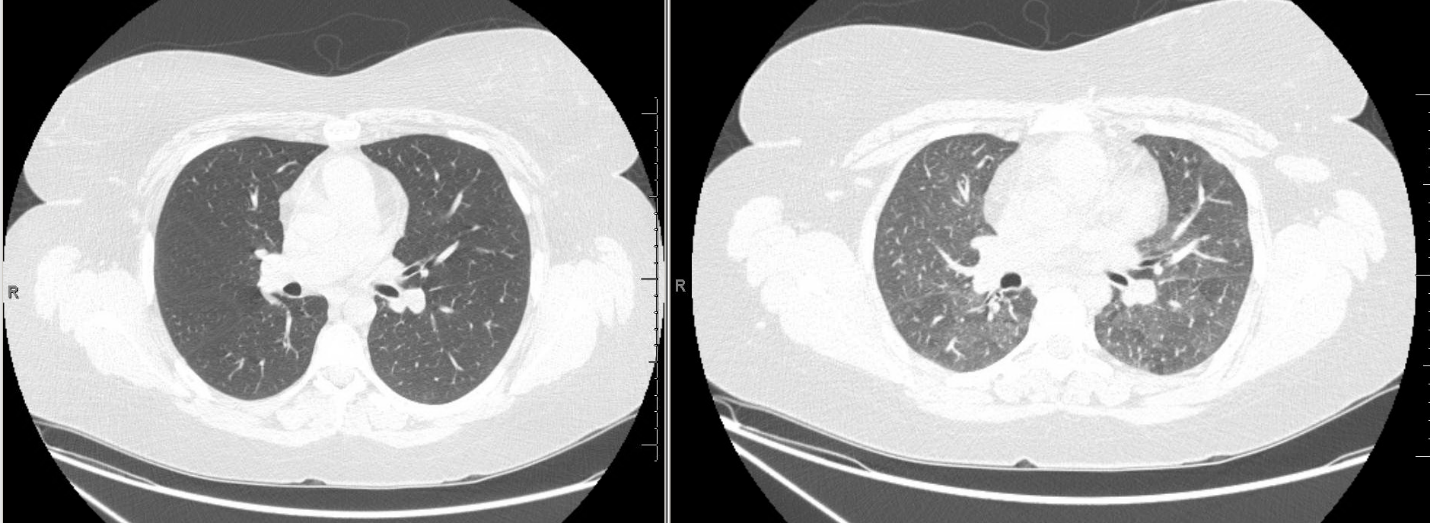
Figures
High resolution CT Imaging
Inspiratory (left) and expiratory (right) images were obtained. Representative images from approximately the same cross sectional level are shown.
References
- Pauwels RA, Löfdahl CG, Postma DS, Tattersfield AE, O'Byrne P, Barnes PJ, Ullman A, for the Formoterol and Corticosteroids Establishing Therapy (FACET) International Study Group. N Engl J Med 1997; 337:1405-11.
- Humbert M, Busse W, Hanania NA, Lowe PJ, Canvin J, Erpenbeck VJ, Holgate S. Omalizumab in asthma: an update on recent developments. J Allergy Clin Immunol Pract. 2014 Sep-Oct;2(5):525-36. PMID: 25213045
- Ortega HG, Liu MC, Pavord ID, Brusselle GG, FitzGerald JM, Chetta A, Humbert M, Katz LE, Keene ON, Yancey SW, Chanez P; MENSA Investigators. Mepolizumab treatment in patients with severe eosinophilic asthma. N Engl J Med. 2014 Sep 25;371(13):1198-207. PMID: 25199059
- Bel EH, Wenzel SE, Thompson PJ, Prazma CM, Keene ON, Yancey SW, Ortega HG, Pavord ID; SIRIUS Investigators. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N Engl J Med. 2014 Sep 25;371(13):1189-97. PMID: 25199060
- Wechsler ME, Laviolette M, Rubin AS, Fiterman J, Lapa e Silva JR, Shah PL, Fiss E, et.al. Bronchial thermoplasty: Long-term safety and effectiveness in patients with severe persistent asthma. J Allergy Clin Immunol 2013; 132: 1295-1302.